Soft X-ray and Energy Storage Science
The pressing demand for high-performance energy storage in today's sustainable energy applications, especially electric vehicles and power grids, requires both conceptual breakthroughs and practical developments of alkali-ion (e.g. Li+ or Na+) batteries [1]. Oxygen (O) redox in battery electrodes is a recent novel conceptual breakthrough beyond conventional transition metal (TM) redox, and has been considered as a promising strategy to enhance energy and power density in storage. Several critical and formidable challenges regarding both the fundamental understanding and practical utilization of O redox remain [2]. While electrochemical and structural characterization have been extensively conducted, the O redox reaction is actually an electronic process. A reliable and direct characterization of O electronic states is thus essential. However, soft X-ray absorption spectroscopy (sXAS), an elemental- and chemical-sensitive probe to 3d TMs widely utilized in investigations of 3d TM redox [4], is subject to intrinsic limitations in characterizing O states in battery electrodes [3].
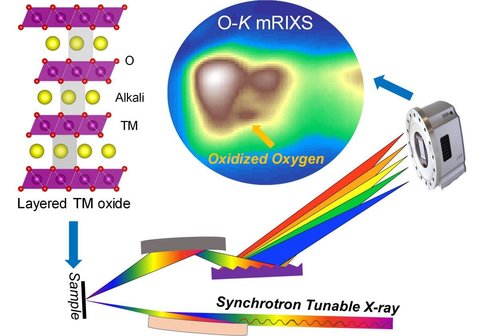
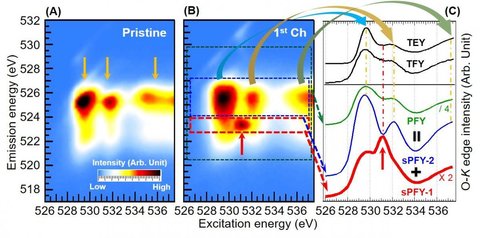
In this project, we study O redox via the advanced mapping technique of resonant inelastic X-ray scattering (mRIXS). We demonstrate the superiority of mRIXS over sXAS on fingerprinting non-divalent O states, and establish a spectroscopic method based on mRIXS to quantitatively analyze O redox. This was done by scrutinizing the O redox process during electrochemical cycling. We several important scientific discoveries regarding its reversibility, performance decay, voltage hysteresis, and kinetics. Lastly, we systematically explored possible approaches for tuning and improving the reversibility of O redox, which brought us a step closer to is use in battery electrodes.
References
[1] Goodenough, J.B. Energy storage materials: A perspective. Energy Storage Materials 1, 158 (2015)
[2] Assat, G., and Tarascon, J.-M. Fundamental understanding and practical challenges of anionic redox activity in Li-ion batteries. Nature Energy 3(5), 373 (2018)
[3] Yang, W., and Devereaux, T.P. Anionic and cationic redox and interfaces in batteries: Advances from soft X-ray absorption spectroscopy to resonant inelastic scattering. Journal of Power Sources 389, 188 (2018)
Selected Publications
[4] J. Wu et al. Modification of transition-metal redox by interstitial water in hexacyanometalate electrodes for sodium-ion batteries. Journal of the American Chemical Society 139(50), 18358 (2017)
[5] J. Wu et al. Dissociate lattice oxygen redox reactions from capacity and voltage drops of battery electrodes. Science Advances 6(6) (2020)
[6] K. Dai et al. High reversibility of lattice oxygen redox quantified by direct bulk probes of both anionic and cationic redox reactions. Joule 3(2), 518 (2019)